Learn
Nuclear Chemistry
Nuclear chemistry, as the name might suggest, is concerned primarily with the changes that occur in the nucleus of an atom. The changes that occur within the nucleus of atoms can be the source of radioactivity and nuclear power.
What two subatomic particles are found in the nucleus of an atom?
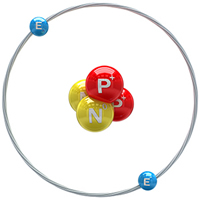
Protons and neutrons in the nucleus are held together by a strong nuclear force. This strong nuclear force is a short-range force that acts as an attractive force for protons and neutrons. When the nucleus is small (contains smaller number of protons and neutrons), the nuclear force is stronger, and the atom is more stable. As the number of protons and neutrons in the nucleus increases, the nuclear force can no longer hold the protons and neutrons as tightly and this is when radioactive decay of the nucleus is more likely to occur.
Open Island of Stability in a new window
Note: The presentation may take a moment to load.
Radioactivity
Radioactivity fullscreen version | Radioactivity text version
Note: The presentation may take a moment to load.
Absorption
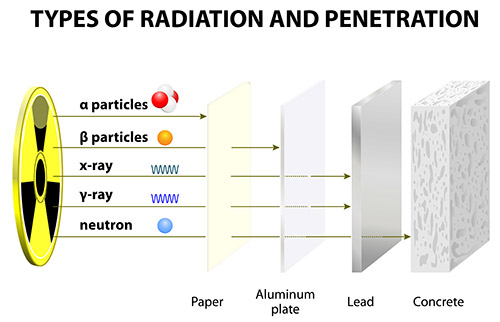
As previously stated, alpha, beta, and gamma radiation can penetrate matter, up to a certain point.
- Alpha radiation, which consists of a helium-4 nucleus, can be readily stopped by something as thin as a sheet of paper.
- Beta radiation, which consists of electrons, can be halted by slightly thicker substances, such as an aluminum plate.
- Gamma radiation can be absorbed by very dense materials. Thick layers of concrete or lead can be used to stop gamma rays from passing through matter. This is due to the density of lead and concrete.