Learn
Radioactivity
Radioactivity, sometimes called radioactive decay, occurs when a large, unstable nucleus loses energy by emitting radiation. A material that spontaneously emits radiation is referred to as radioactive.
So what does it mean to emit radiation? Well, radiation is the emission of energy or the subatomic particles carrying energy.
Radioactive substances are actually detected by measuring the nuclear radiation they give off. As the nucleus of a radioactive substance decays (or breaks down) they release charged particles and energy. The emission of particles and energy in this process is called nuclear radiation.
There are three basic types of nuclear radiation:
- Alpha
- Beta
- Gamma rays
Both alpha and beta radiation are composed of particles, whereas gamma radiation is composed of electromagnetic radiation. You will learn a little more about the three types in the next sections.
Alpha Particles
An alpha particle (α) consists of two protons and two neutrons bound together and emitted from the nucleus during some kinds of radioactive decay. It is the same as a helium-4 nucleus. An alpha particle can be written as ![]() .
.

Generally, alpha particles occur in very heavy nuclei where the number of protons and neutrons need to be reduced in order to increase the stability of the nucleus. Because these particles interact with other matter repeatedly, they transfer energy to their surrounding very quickly as they travel through solids, liquids, and gases.
Alpha particles are the least penetrating form of nuclear radiation. A simple sheet of paper will stop most alpha particles.
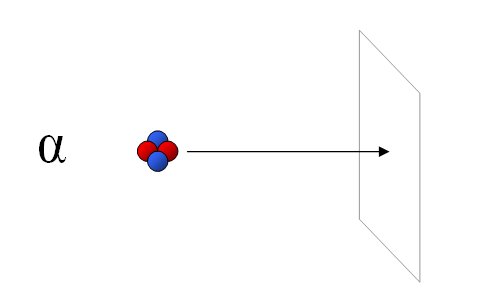
Alpha particles can be stopped by a substance as thin as a sheet of paper.
Beta Particles
A beta particle (β) is a high-energy electron emitted from the nucleus when a neutron decays into a proton. A beta particle can be written as 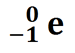 .
.
Beta particles are caused by the weak nuclear force. The weak nuclear force is a short-range force that is stronger than gravitational force, yet weaker than the other two forces (strong force and electric force). When the neutron to proton ratio is too high, the weak nuclear force causes neutrons to decay into protons.
Beta particles are much more penetrating and faster than alpha particles. It takes at least a thin sheet of aluminum foil to stop beta particles.
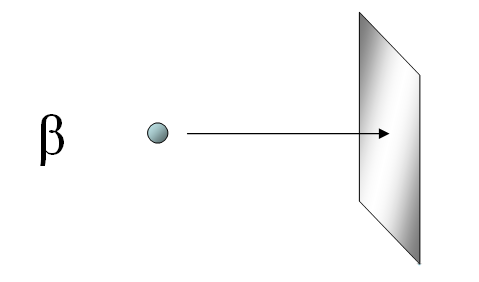
Beta particles can be stopped by thin sheet of aluminum foil.
Gamma Rays
Gamma rays (γ) are high-energy electromagnetic waves that are emitted from the nucleus.
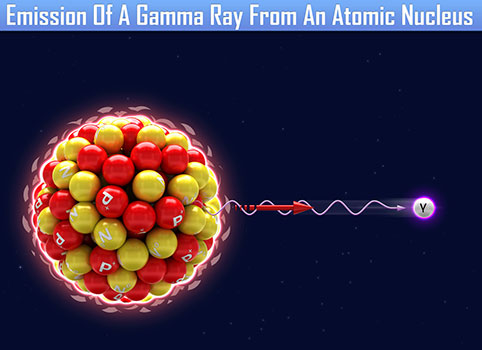
All forms of radiation can penetrate matter, up to a certain point. Thick layers of dense materials, such as several feet of concrete or lead, are needed to prevent gamma rays from moving through matter.
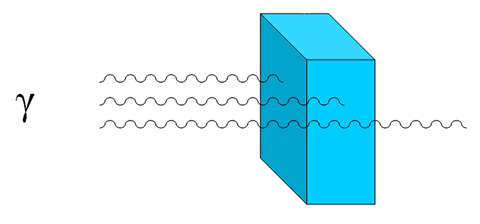
Gamma particles can penetrate thicker substances than alpha and beta particles but can be stopped by thick materials like layers of concrete.